

The research group “Neurological B cell Immunology” is focused on the role of B cells in neuro-inflammatory diseases including multiple sclerosis (MS) and neuromyelitis optica spectrum disorder (NMOSD). Whereas aquaporin (AQP4) autoantibodies could be clearly linked to NMOSD pathophysiology, the exact role of B cells in multiple sclerosis still remains unknown. However, B cell depleting therapies have been shown to be highly effective in multiple sclerosis indicating, that B cells play an important role in MS pathogenesis. Our primary aim is to further understand the specific functions of B cells in multiple sclerosis and other autoimmune diseases. Besides neuro-inflammatory diseases, we are also interested whether B cells are involved in the pathophysiology of primarily non-inflammatory neurological diseases (e.g. ischemic stroke and cerebral tumors) in terms of a secondary reaction after disruption of the physiological tissue architecture.
Immunoglobulin repertoire analysis on transcriptome level
By means of next generation mass sequencing, up to several million immunoglobulin (Ig) sequences can be assessed on transcriptome level. By using a bioinformatics approach, these Ig repertoires can be further analysed in regard to e.g. Ig subclasses, diversity and clonality. In addition, lineage analysis of clonally related B cells allows a detailed characterization of maturation and migration processes.
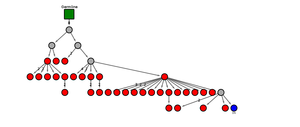
Example for a B cell maturation tree with a cerebrospinal fluid B cell (blue) connecting to a peripheral blood B cell cluster (red dots). By this lineage analysis, B cell migration and maturation processes between different compartments can be studied in detail.
Immunoglobulin repertoire analysis on proteome level
The immunoglobulin protein fraction is first separated from other proteins, digested to obtain Ig peptides and further analysed by means of mass spectrometry. The resulting Ig peptides are then aligned to the Ig transcriptome repertoires in specific regions in order to understand the dynamics of B cell maturation and actual antibody production.
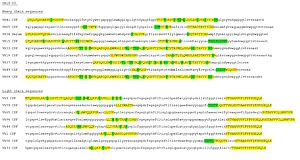
Overlap between Ig transcriptome and Ig proteome: Ig peptides (marked in yellow) are aligned to Ig transcriptome sequences and checked for specificity (mutations to germline, marked in green). By this approach the dynamics between circulating B cells and actual antibody production can be characterized.
Production of recombinant antibodies
Paired immunoglobulin heavy and light chains can by assessed by using a single cell approach in order to produce recombinant antibodies, originating from specific B cells. Those antibodies can then be tested for their specific targets and analysed in regard to functional properties.

07071
86703
Ruschil C, Kemmerer CL, Beller L, Gabernet G, Kowarik MC. Next Generation Sequencing of Cerebrospinal Fluid B Cell Repertoires in Multiple Sclerosis and Other Neuro-Inflammatory Diseases-A Comprehensive Review. Diagnostics (Basel). 2021 Oct 11;11(10):1871.
Kemmerer CL, Schittenhelm J, Dubois E,Neumann L, Häsler LM, Lambert M, Renovanz M, Kaeser SA, Tabatabai G, Ziemann U, Naumann U, Kowarik MC. Cerebrospinal fluid cytokine levels are associated with macrophage infiltration into tumor tissues of glioma patients. BMC Cancer. 2021 Oct 15;21(1):1108.
Ruschil C, Gabernet G, Lepennetier G, Heumos S, Kaminski M, Hracsko Z, Irmler M, Beckers J, Ziemann U, Nahnsen S, Owens GP, Bennett JL, Hemmer B, Kowarik MC. Specific Induction of Double Negative B Cells During Protective and Pathogenic Immune Responses. Front Immunol. 2020 Dec 18;11:606338.
Kowarik MC, Astling D, Lepennetier G, Ritchie A, Hemmer B, Owens GP, Bennett JL. Differential Effects of Fingolimod and Natalizumab on B Cell Repertoires in Multiple Sclerosis Patients. Neurotherapeutics. 2020 Nov 30. Epub ahead of print.
Kemmerer CL, Pernpeintner V, Ruschil C, Abdelhak A, Scholl M, Ziemann U, Krumbholz M, Hemmer B, Kowarik MC. Differential effects of disease modifying drugs on peripheral blood B cell subsets: A cross sectional study in multiple sclerosis patients treated with interferon-β, glatiramer acetate, dimethyl fumarate, fingolimod or natalizumab. PLoS One. 2020 Jul 27;15(7):e0235449.
Lepennetier G, Hracsko Z, Unger M, Van Griensven M, Grummel V, Krumbholz M, Berthele A, Hemmer B, Kowarik MC. Cytokine and immune cell profiling in the cerebrospinal fluid of patients with neuro-inflammatory diseases. J Neuroinflammation. 2019, 16(1):219.
Kowarik MC, Astling D, Gasperi C, Wemlinger S, Schumann H, Dzieciatkowska Monika, Ritchie AM, Hemmer B, Owens GP, Bennett JL. CNS Aquaporin-4 specific B cells connect with multiple B cell compartments in neuromyelitis optica spectrum disorder. Ann Clin Transl Neurol. 2017, 4(6):369-380.
Kowarik MC, Dzieciatkowska M, Wemlinger S, Ritchie AM, Hemmer B, Owens GP, Bennett JL. The cerebrospinal fluid immunoglobulin transcriptome and proteome in neuromyelitis optica reveals central nervous system-specific B cell populations. Journal of Neuroinflammation. 2015, 12:19.
Kowarik MC, Soltys J, Bennett JL.The treatment of neuromyelitis optica. Journal of neuro-ophthalmology. 2014, 34(1):70-82.
Kowarik MC, V Grummel, S Wemlinger, D Buck, M S Weber, A Berthele, B Hemmer. Immune cell subtyping in the cerebrospinal fluid of patients with neurological diseases. Journal of Neurology. 2013, 261(1):130-43.
Kowarik MC, Cepok S, Sellner J, Grummel V, Weber MS, Korn T, Berthele A, Hemmer B. CXCL13 is the major chemokine for B cell recruitment and B cell response in the CSF during neuroinflammation. Journal of Neuroinflammation. 2012, 9:93.
Kowarik MC, Einhäuser J, Jochim J, et al. Impact of the COMT Val108/158Met polymorphism on the mu-opioid receptor system in the human brain: mu-opioid receptor, met-enkephalin and beta-endorphin expression. Neuroscience letters. 2012, 506(2):214-9.
Kowarik MC, Pellkofer HL, Cepok S, Korn T, Kümpfel T, Buck D, Hohlfeld R, Berthele A, Hemmer B. Differential effects of fingolimod (FTY720) therapy on immune cells in the cerebrospinal fluid and blood of multiple sclerosis patients. Neurology. 2011, 76(14):1214-21.
Kowarik M, Onofri C, Colaco T, Stalla GK, Renner U. Platelet-derived growth factor (PDGF) and PDGF receptor expression and function in folliculostellate pituitary cells. Exp Clin Endocrinol Diabetes. 2009, 118(2):113-20.
Close cooperation with PD Dr. M. Krumbholz and staff.
Prof. U. Naumann, Molecular Neurooncology, Hertie Institut für klinische Hirnforschung
Dr. S. Nahnsen, Zentrum für Quantitative Biologie (QBiC)
Dr. S. Autenrieth: Flow Cytometry Core Facility, Universitätsklinikum Tübingen
Dr. N. Casadei: Sequencing Core facility, Universitätsklinikum Tübingen
International collaborations:
Jeffrey Bennett und Greg Owens, Department of Neurology, University of Colorado, USA














